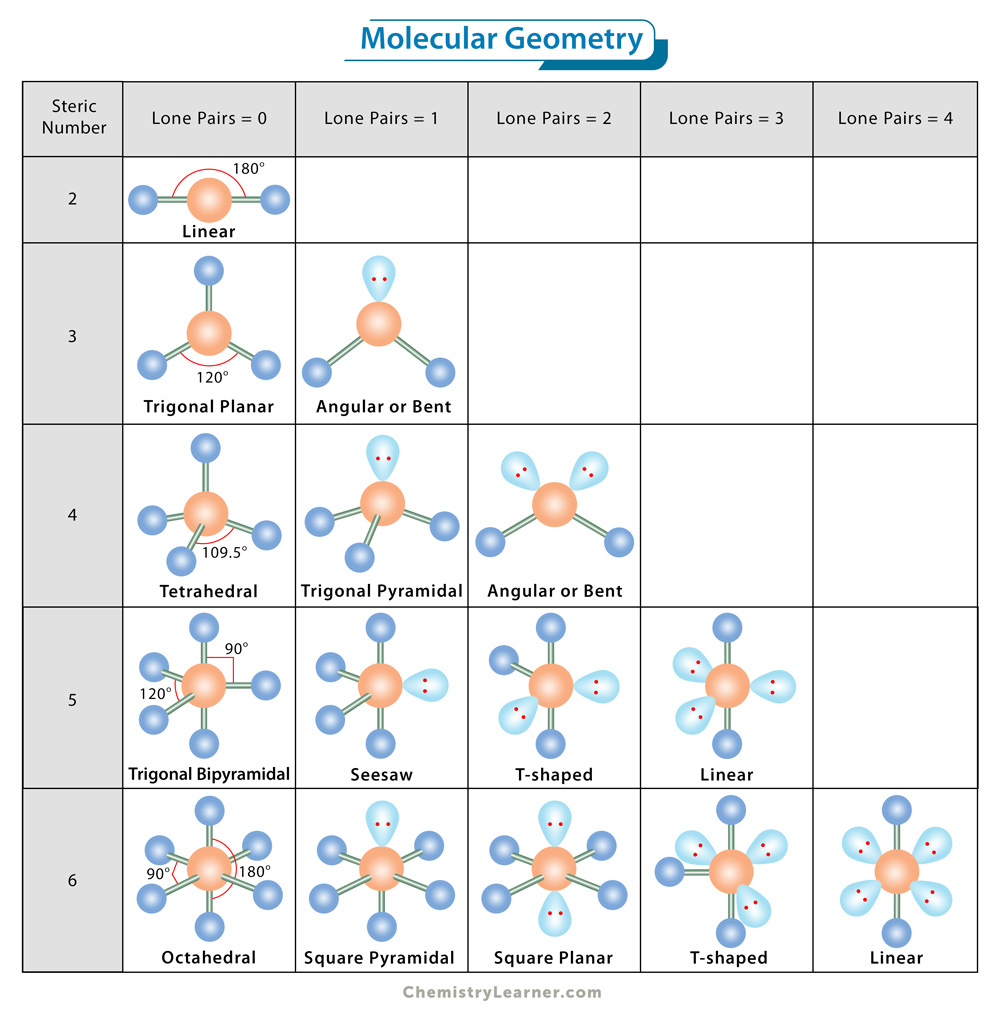
Differences: Electron Geometry and Molecular Geometry There are also 2 single bond pairs, so the resulting shape is bent. So there are 4 electron groups arranged in a tetrahedral shape. Here, oxygen (O) is the central atom with 6 valence electrons so it requires 2 more electrons from 2 hydrogen atoms to complete its octet. Unlike electron geometry, it is predicted by considering only electron pairs. For small molecules, the molecular formula and a table of standard bond lengths and angles may be all that is required to determine the geometry of the molecule. The geometry of a molecule is usually described in terms of bond lengths, bond angles, and torsional angles. Understanding the molecular geometry of a compound helps determine the reactivity, polarity, color, phase of matter, and magnetism. It simply refers to the three- dimensional arrangement or structure of atoms in a molecule. Molecular geometry is used to determine the shape of a molecule. This means there are a total of 8 electrons around carbon and there are no single bonds, so the number of lone pairs here is 0. Here, the central atom is carbon with 4 valence electrons and 4 hydrogen share electrons with 1 carbon to form 4 covalent bonds. Let’s take an example of methane, the chemical formula of which is CH4. Because electrons are always in constant motion and their paths cannot be precisely defined, the arrangement of the electrons in a molecule is described in terms of an electron density distribution. Electron pairs are defined as electrons in pairs or bonds, lone pairs, or sometimes a single unpaired electron. The term electron geometry refers to the name of the geometry of the electron pair/groups/domains on the central atom, whether they are bonding electrons or non-bonding electrons. Although these terms sound similar, there are fundamental differences between them that are important to understand. 
These concepts are related to the arrangement of atoms in a molecule and the distribution of electrons around those atoms. 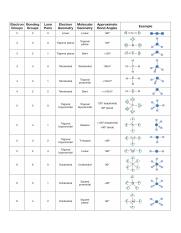
In this case an extra step is needed to to translate from electron pair geometry to the final molecular geometry, since only the positions of bonded atoms are considered in molecular geometry.Electron geometry and molecular geometry are two concepts that are important in the field of chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
